7 Smart Ways to Prevent Diagnostics System Development Projects from Failing

Posted by:
Michael Collasius
Published on:
Jun 24, 2019
Diagnostic system development projects can be more prone to failure than you would think. Despite the eye-watering amounts of time and money that flow into these projects, relatively simple aspects are often overlooked. Whether you are a start-up or a large corporate organization, here are my top 7 prerequisites for successful diagnostic system development.
1. Have clearly defined project goals
The Food and Drugs Administration (FDA) asks for the “intended use” of a diagnostic product for a very good reason. This defines why a product is being developed and who will benefit from it. However, the reality is often very different.
Let's say someone has a cool idea for a “disruptive” technology. The excitement is high, a decision is made and the project gets off to a great start. Everyone seems to be clear on what needs to be done and a large number of people work on something that relates to the original idea. So far so good. However, when things become more specific, the first problems are encountered. Some of these problems are addressed, others are not. The scope creeps again and again as new ideas are incorporated. The amount of work increases, the costs spiral out of control and project progress comes to a standstill.
The solution to prevent this happening is surprisingly simple. Make sure the project goal is clearly defined, and that you have actionable objectives on how to achieve these goals. All stakeholders should then understand and agree on these goals. In many cases it can also be helpful to define what is out of scope. Here you can download a helpful FDA Checklist.
2. Understand your customer's requirements
Surprisingly, the customer is often left out of the product-development equation. Either because we think we know what the customer wants, someone has a better idea of what the customer needs, or there is no time to ask the customer as the new product is needed urgently. This approach is fraught with danger. No matter how successful the development, a product will fail after launch if nobody wants it or sales lag behind the original business case.
Talking to the customer and understanding their requirements is will save you both time and money. There is a very good reason why requirements management is a separate discipline: this ensures that customer needs are fully understood and critical requirements are identified. Resolutely following a list of all aspects customers may have ever wished for makes a product expensive and possibly still misses out on what the customer really wanted.
3. Get the specifications right
Now that customer needs are clear, there is an infinite number of options and ways of implementing these requirements. Since everyone is on the same wavelength and timelines are short, the team immediately gets cracking and defines the solution, but the specifications are loosely defined or, even worse, not at all. This is the perfect recipe for disaster, as development will be a never-ending process or will not deliver what was intended. The other risk scenario is overly ambitious specifications. With this scenario the timelines will drag on, while development and manufacturing costs for the final product go through the roof.
Deriving the specifications from the requirements is time well spent and saves time in the long run. Make sure you know how to measure specification achievement and develop appropriate measurement tools for this.

Well-defined, achievable specifications are a prerequisite for a profitable diagnostic testing business after product launch.
4. Invest in project management (and don't cut corners)
Successful diagnostic platform development largely depends on disciplined implementation of the customer requirements and product specifications defined in the project goal. The development process is the collective, documented experience of an organization. It gives guidance on what needs to be done, puts actions into the right order and aligns activities across multiple disciplines. Although this sounds simple, people often “cut processes short” for faster timelines or “leaner, less cumbersome” procedures. Shortcuts will almost inevitably lead to longer timelines, higher costs, undermine the approval process and can ultimately lead to project failure. In case you are running out of resources, consider outsourcing parts of your development processes to external experts.
5. Assemble an experienced team
Being unaware of what you don’t know poses the biggest risk to diagnostic system development projects.
A range of biology and engineering disciplines need to be aligned, pushing costs into several hundred million US$. Typical system development tasks include knowledge on medical needs and regulatory requirements, applications development, chemistry, plastic disposables, mechanics, electronics and computer software.
Dangers arise when all these disciplines work independently, sometimes up to two-thirds of the project time. The timelines, scope and cost are still within the project plan, but this can go awry when the individual pieces of the puzzle are put together to form an integrated diagnostic system. Individual components are usually not ready at the right time and fitting them together reveals unexpected challenges.
An experienced team is aware of the risks, is familiar with these traps and has learned to identify the critical elements and take appropriate measures. If your organization does not have appropriate experience, seek professional advice. Assemble an independent advisory board and take their advice seriously.
6. Assign responsibilities clearly
You would think this is implicit, but in many cases responsibilities are not clearly assigned and documented. Smaller organizations tend not to assign responsibilities within projects, assuming the hierarchical structure in the organization will take care of everything. A misconception that results in double work, tasks fall through the cracks, potential problems became apparent too late, money is lost and, in the worst case scenario, jeopardizes the company's solvency. Even in very small organizations, assigning responsibilities well structures the project and boosts transparency.
In larger organizations the situation is a little different. Organizational deficits or a complex political environment can prevent clear assignment of responsibilities, putting projects at risk. Clear role descriptions and assignments consistent with competencies, decision power and appropriate work package breakdowns are the most important building blocks for a successful diagnostic system project.
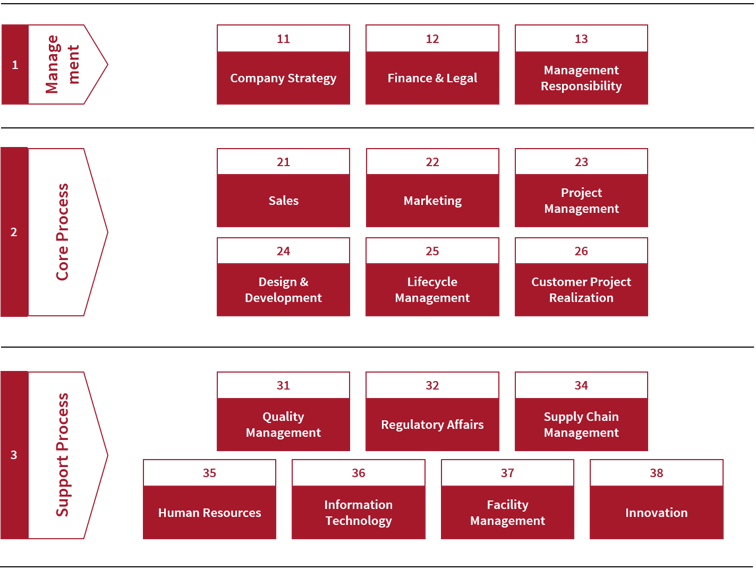
HSE guideline for assigning project process responsibilities.
7. The most underestimated role? The project sponsor
While it is quite common to have a project manager, the role of a project sponsor is relatively unknown and is often seen as unnecessary or playing a secondary function. The project sponsor is the most senior stakeholder manager defending the project within the organization and ensuring the business goals of the project.
Although a project sponsor is not assigned in many cases, they have a vital function: this is the person who ordered the project, possibly as a delegate of an executive team, and pays for it. While the boundaries of a project manager are set by scope, time and cost, the project sponsor can make appropriate decisions if the project faces obstacles that cannot be resolved within its scope.
In conclusion
Although the above building blocks don't cover all aspects of a successful system development project, they form a great foundation. Diagnostics development projects in particular are complex undertakings with many unknowns, even when the project is planned meticulously.
As noted by our CTO Konstantin Lutze on the success of interdisciplinary automation projects, clients and stakeholders need a unified vision to prevent project failure. These basics are so important that we offer you a free assignment template to keep your team on the same page.
Are you facing difficulties during your system development process? Contact us and we will share with you our Project Management QMS Procedures for development, quality and life cycle management standardization.

